In the fields of modern fine chemicals and cultural heritage conservation, selecting appropriate consolidants and coating materials presents a highly challenging task. This is particularly true for composite objects containing both organic components (such as wood) and metals (such as bronze), where material compatibility and chemical stability directly determine the longevity of the cultural artifacts. This article delves into Polyvinyl butyral (PVB)—specifically Eastman Butvar B-98—examining its chemical structure, industrial properties, and anti-corrosion performance in harsh environments.

1 Chemical Structure and Polymerization Characteristics of PVB Resin
PVB is not a simple homopolymer; rather, it is a terpolymer composed of three distinct monomers. It is synthesized through the reaction of polyvinyl alcohol (PVOH) with butyraldehyde under specific conditions.
1.1 Terpolymer Components
The physical properties of the Butvar product series (such as B-98) are determined by the proportions of the following three functional groups:
Polyvinyl butyral (PVB): Provides hydrophobicity and mechanical strength.
Polyvinyl alcohol (PVOH): Residual hydroxyl groups provide adhesion and solubility.
Polyvinyl acetate (PVAC): Controls the viscosity of the polymer.
Taking Butvar B-98 as an example, its typical composition consists of 80% PVB, 18–20% PVOH, and 0–2.5% PVAC. This specific ratio endows the material with excellent mechanical strength, flexibility, and solubility in non-toxic solvents.
1.2 Physicochemical Parameters
Studies indicate that PVB demonstrates superior performance compared to acrylic resins and PVAC in the context of wood consolidation; furthermore, virtually no shrinkage or expansion is observed during the treatment process. Additionally, it possesses a relatively high glass transition temperature (Tg), and its viscosity can be precisely controlled by adjusting the solvent carrier.
2 Applications of Butvar B-98 in Industrial and Protective Fields
One of the most significant industrial applications of PVB resin is its use as a coating for metals. Its exceptional adhesion and chemical stability make it a preferred choice for use in a wide variety of environments.
2.1 Reinforcement of Composite Materials: In the restoration of an 8th-century BC bronze-decorated wooden stand excavated at Gordion, Turkey, researchers utilized a 10% solution of Butvar B-98 (using an ethanol/toluene solvent mixture with a ratio of 60:40) reinforced using a solution of (Ethanol/Toluene). In this specific case, Butvar was employed to consolidate fragile, desiccated boxwood, leveraging its exceptional penetrative properties and structural support capabilities.
2.2 Use of Auxiliary Chemicals: In practical applications, other chemical agents are often used in conjunction with Butvar to further enhance the corrosion resistance of metals:
BTA (Benzotriazole): Used for the pretreatment of metal surfaces to inhibit chemical reactivity.
Paraloid B-72: Applied as an additional coating to provide a dual layer of protection.
3. In-Depth Experimental Analysis of Butvar's Corrosivity Toward Bronze
For a considerable time, the conservation community has harbored concerns regarding whether Butvar releases volatile organic acids (such as butyric acid) that could subsequently corrode metals. To address this issue, Queen's University conducted accelerated aging experiments on Butvar B-98 using a modified Oddy test.
3.1 Experimental Methodology and Equipment
Researchers suspended bronze test coupons—composed of 6% tin (Sn) and 94% copper (Cu)—within sealed containers and subjected them to aging for one month in a high-humidity environment maintained at 60°C.
The experiment utilized a range of precision analytical techniques:
XRD (X-ray Diffraction): To analyze the composition of the corrosion products.
FTIR (Fourier-Transform Infrared Spectroscopy): To analyze the chemical changes occurring in the Butvar film before and after aging.
Cold Extraction pH Test: To measure the acidity/alkalinity of the dried film.
3.2 Identification of Corrosion Products
The experiments revealed that corrosion occurred on the bronze test coupons regardless of whether they were in contact with Butvar. XRD analysis confirmed that the resulting corrosion products consisted primarily of:
Tenorite (CuO): Indicating that an oxidation reaction had taken place.
Atacamite (Cu₂ClOH₃) and Clinoatacamite (Cu₂OH₃Cl): These are the primary agents responsible for "bronze disease," a condition typically triggered by the presence of chloride ions in the environment.
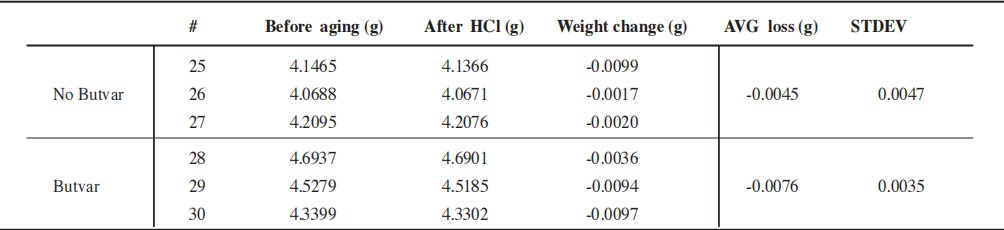
3.3 Data Comparison
According to the experimental records, the difference in average weight loss between the bronze coupons exposed to Butvar and those not exposed fell within the range of the standard deviation; this result demonstrates that Butvar did not accelerate the corrosion process.
4. Assessment of Photothermal Degradation and Long-Term Stability
The photo-oxidative degradation of PVB is influenced by its glass transition temperature (Tg). At temperatures exceeding the Tg, the polymer chains are prone to cross-linking; conversely, in normal environments below the Tg, the primary degradation mechanism involves chain scission, which helps to preserve the polymer's solubility. The volatile byproducts generated during degradation consist primarily of butanal and water.
Generation of Volatile Acids
Although degradation does result in the formation of butyric acid, the quantity produced is negligible. Experimental data indicate that after 455 hours of exposure to UVA radiation, only one mole of acid is generated for every 70 moles of aldehydes released.
Service Lifetime Prediction
Based on estimates, under typical museum lighting conditions (approximately 23 lux), PVB materials exhibit an induction period—the time elapsed before significant weight loss or a shift in degradation mechanism becomes apparent—that may extend up to 113 years.
In summary, experimental results demonstrate that under accelerated aging conditions, Butvar B-98 does not release volatile substances into the surrounding environment in quantities sufficient to cause corrosion in bronze. Following testing, the material's pH remained stable within the range of 6.6 to 7.0, falling well within the safe threshold. For professionals in the chemical coatings industry and conservation specialists alike, Butvar B-98 remains a highly efficient and stable choice for the treatment of wood-metal composite artifacts. Nevertheless, given the inherent non-linear discrepancies between accelerated aging experiments and actual long-term environmental conditions, continuous environmental monitoring (specifically, the control of temperature and relative humidity)—coupled with the concurrent use of corrosion inhibitors such as BTA—remains the optimal best practice.
Website: www.elephchem.com
whatsapp: (+)86 13851435272
E-mail: admin@elephchem.com